For months now, your Beetles in the Bush host, Ted, has been nudging me to blog, in the end resorting to offering me a guest blogger gig at BitB. Given this golden opportunity, I’ve decided to utilize my web-logging debut to introduce my favorite insects, the fabulous Formicidae. First, a disclaimer: I have not mastered ant photography, and so will rely on the undisputed king of ant photographers, Alex Wild, through links to his numerous, unexcelled images.
Since about age 5, I can remember being interested in virtually anything living, but especially in small, active creatures. From the beginning, I have had a particular attraction to ants. With some notable exceptions, and aside from the pulchritudinous feature of their svelte waists, ants aren’t what most folks would call pretty, but they are — How else to say it? — just plain “cool”!
First, who are they and where do they come from? Ants constitute a single family, Formicidae, within the insect order Hymenoptera, so their relatives are wasps, bees, sawflies, horntails, gall wasps, and a vast array of small parasitic wasps that are mostly unappreciated except by specialists who study them. Within Hymenoptera, the ants are considered to belong to the superfamily Vespoidea, along with hornets, paper wasps, potter wasps and other solitary relatives. The evidence at present indicates the first animals we would call ant had diverged from their common ancestry with these other stinging wasps some time in the Cretaceous, 130 million years ago, more or less. Ants are classified in a varying number of subfamilies, currently at about 20. Fossils in amber up to 100 million years old represent early members of several modern subfamilies, and a few extinct groups. Most of us in the Northern Hemisphere Temperate Zone are familiar only with the big two subfamilies, Formicinae (carpenter ants, weaver ants, honey ants, etc.) and Myrmicinae (fire ants, harvester ants, leaf-cutter ants, etc.). In much of North America, folks may also be familiar with an abundant member of another subfamily, Dolichoderinae, namely odorous house ants, which frequent our gardens, kitchen counters, wall spaces, and even electrical outlets, especially in spring.
Ants are a conspicuous and often dominant presence in the World of the Little (or, what Piotr Nascrecki, in one of my favorite books, calls the “Smaller Majority” ). It is difficult for any observant person to sit still, outdoors in good weather, and not begin to see ants doing what ants do. They scurry about singly, in pairs or threesomes or foursomes, or in long lines, or columns. Our notice may be further piqued by their habit of transporting sundry bits of biomass or mineromass (pebbles, etc.). Often this is just taking out the inedible food waste, or sawdust or soil excavated while expanding or remodeling their nests. Less visibly, because more diffusely in space, ants carry a variety of items from foraging to their nests to provide nutrition for their colonies, or to add mass or create functional structure to their nests (to create better drainage, to provide incubation space for developing brood, and in some desert ants, to capture dew). In one of the most spectacular examples of ants transporting things, the so-called “slave-making” ants carry home the mature brood of a related species, these young ants later maturing in the brood-robbers’ nest to become its work force!

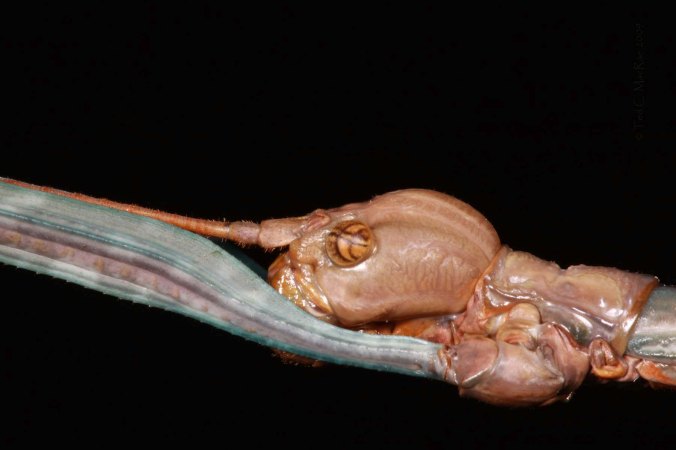
Shiny red workers of Polyergus lucidus return with pupae pillaged from a nest of Formica incerta several meters away. Two brown and differently proportioned workers of the latter that matured from raids earlier in the life of this Polyergus colony may be seen at the right of the photo.
Perhaps, not so widely known is that most of what most ants carry home is not some large, heavy particle in their mandibles, but rather is liquid carried in an expansible section of their esophagus called the crop. Because of the fine diameter of their gullets, adult ants cannot eat anything other than the most minute solid particles (e.g., pollen grains, loose cells from their prey). Solid items may be cut up to feed to the legless, pale larvae, or the larvae may even be placed directly upon the killed prey to bite into it and feed on their own, using their flexible “necks”. Adult ants get pre-digested food in return, in the form of glandular secretions loosely termed saliva, but which may be either a glandular secretion from the larva itself or simplify pre-liquefied flesh of prey lapped up from the larva’s messy eating. In some lineages, known as Dracula ants, adults actually “bleed” the larvae through rapidly healing wounds made at particular locations on the larval exoskeleton.
Okay, I need to get back to my regular work, so let’s bring this home (to winter in the United States). Many of us are now in the dead of winter, or so it would seem. But, on sunny days, sap is beginning to flow upward in maple and other trees, and one ant species may actually be seen, creeping slowly through the woods, in search of dead arthropods and earthworms, or perhaps some sweet sap oozing from a sapsucker wound in a tree. This is Prenolepis imparis, sometimes called “winter honeypot ant”. This is a partial misnomer. While foragers may indeed fill their crops to over-full with sweet sap or honeydew, the very bloated “honeypots” in the deep nests of this ant are in fact, fat pots, having converted their food to whitish body fat. This is later converted to a glandular secretion that serves as food for developing larvae. These ants are likely to be seen anywhere near where oaks of just about any species grow, and the where the soil is moist but well-drained. Look for these shiny little dark brown ants during your walks in the woods, on the sunny days that are sure to increase in number and warmth in the coming months.
Copyright © James C. Trager 2010










 Those of you who have followed this blog for any length of time have likely noticed fairly regular participation in the comments sections by one James C. Trager. Occasionally irreverent and always articulate, his informed quips are among those that I have enjoyed the most. One can surmise from James’ comments that he knows a thing or two about entomology himself, but to say this would be an understatement! Like me, James is a passionate entomologist whose scientific interests take him deep into many related fields of natural history study. Unlike me, James is a formally trained insect taxonomist, specializing in ants (family Formicidae). He has conducted numerous biogeographical and systematic studies on this group, much of it in the southeastern U.S. (list of
Those of you who have followed this blog for any length of time have likely noticed fairly regular participation in the comments sections by one James C. Trager. Occasionally irreverent and always articulate, his informed quips are among those that I have enjoyed the most. One can surmise from James’ comments that he knows a thing or two about entomology himself, but to say this would be an understatement! Like me, James is a passionate entomologist whose scientific interests take him deep into many related fields of natural history study. Unlike me, James is a formally trained insect taxonomist, specializing in ants (family Formicidae). He has conducted numerous biogeographical and systematic studies on this group, much of it in the southeastern U.S. (list of