A few weeks ago I received an email from Kyle Schnepp, an entomology student at Purdue University. Kyle has taken on the rather ambitious project of developing an illustrated key to the Buprestidae of eastern North America, for which he has been spending the past year acquiring material for photographs.
During his examination of specimens in the Field Museum of Natural History, Kyle came across two examples of an extraordinarily rare species of Buprestidae, Agrilus audax Horn. Although described more than 100 years ago from specimens collected in Texas (Horn 1891), few records have been published in the years since. Chamberlin (1926) reported the species also from Arizona and Illinois but without further details, causing Fisher (1928), in his revision of the genus (woefully out-of-date now, but still the only comprehensive resource for identifying the North American species), to regard at least the Illinois record as probably erroneous (common for many of Chamberlin’s records). The first undisputed report of this species from outside of Texas was by Josef Knull (1934), who reported the species emerging from living, wind-thrown branches of slippery elm (Ulmus rubra) collected in western Missouri. More than half a century would pass before the species would turn up again – first in Oklahoma (Nelson and MacRae 1990) and twice again in Missouri through the efforts of Gayle Nelson and myself (MacRae 1991, MacRae and Nelson 2003). All but one of these specimens were beaten from bur oak (Quercus macrocarpa).
While the rarity of this species makes Kyle’s find significant enough, there is an even more significant – and interesting – aspect to his discovery. Both of the specimens, one male and one female, were collected in Ohio, which is a rather extraordinary geographical range extension. Additionally, the specimens were collected by none other than Josef Knull. To students of North American Buprestidae, the name Josef Knull is as familiar as Carl Linnaeus, Charles Darwin, or Thomas Say. A Professor of Entomology at The Ohio State University from 1934-1962, Knull published nearly 200 papers on the taxonomy, biology, and distribution of these and other families of beetles (Davidson and Bellamy 2002). Although he lacked a Ph.D., he was an indefatigable collector and describer of beetles – to his fellow colleagues and students, he was known as “Professor” or “Doctor” as a show of respect. He spent many of his summers traveling through the southwestern U.S. with his wife Dorothy Knull (herself an entomologist specializing in leafhoppers), and by the time he died in 1975 he had described 233 species and subspecies of beetles (as well as one species of Fulgoridae). He was, and is, an icon among North American beetle collectors.
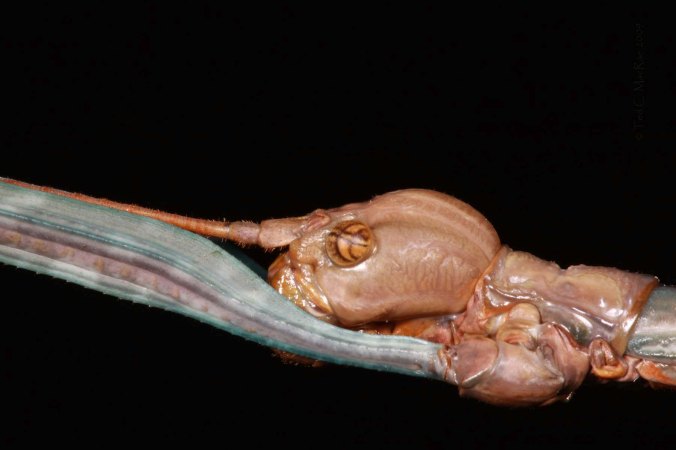
Curiously, Knull did not recognize these specimens for what they were, instead identifying them as the similar and much more widespread species, Agrilus vittaticollis. Curious, because Knull collected these specimens in 1949 and 1953 – after first reporting the species in Missouri. Agrilus audax belongs to a small group of species that look very similar to each other by way of their large size and striking coloration – black elytra and a red pronotum with a densely pubescent median channel. Agrilus vittaticollis is the most common of these (though still not as commonly encountered as many other species in the genus), and the much less common A. benjamini also belongs to this group. Kyle had sent me the above photo in an attempt to confirm their identity, but true confirmation would require examination of characters of the face and ventral surface. Kyle quickly took additional photographs of these characters and sent them to me – they are shown below and leave no doubt as to the identity of these specimens.

Agrilus audax frons is moderately depressed and uniformly pubescent (deeply depressed & pubescent only on lower half in A. benjamini).

Agrilus audax male sternite – the deep, smooth, elongate depression is diagnostic (A. benjamini males have only an obsolete depression).
Finding a new state record buprestid in Ohio – the land of Knull – based on specimens collected by Knull himself is nothing short of remarkable (almost like proving E. O. Wilson wrong¹). The occurrence of A. audax in Ohio also lends some credibility to Chamberlin’s record of the species in Illinois. Kyle is graciously allowing me to include these new records in a forthcoming publication; my thanks to him for this and also for allowing me to use his fine photographs in this post. Kyle did also mention that these were the only misidentified specimens he saw in the Knull collection at the Field Museum of Natural History. For those interested in acquiring reprints of Knull’s papers, pdfs of the 50 papers he published in the Ohio Journal of Science may be found at this link.
¹ The title of this post is a play on the title of a recent post by Alex Wild at Myrmecos. No true disrespect is intended to Josef Knull, who’s legacy (with the possible exception of his frustratingly vague label data) is rightfully held in high regard by all who knew him or know of his work.
REFERENCES:
Chamberlin, W. J. 1926. The Buprestidae of North America, exclusive of Mexico, a catalogue including synonymy, bibliography, distribution, type locality and hosts of each species. W. J. Chamberlin, Corvallis.
Davidson, J. M., and C. L. Bellamy. 2002. The entomological contributions of Josef Nissley Knull (1891-1975). Zootaxa 37:1-24.
Horn, G. H. 1891. The species of Agrilus of Boreal America. Transactions of the American Entomological Society 18:277-366.
Knull, J. N. 1934. Notes on Coleoptera, No. 4. Entomological News 45(10):207-212.
MacRae, T. C. 1991. The Buprestidae (Coleoptera) of Missouri. Insecta Mundi 5(2):101–126.
MacRae, T. C., and G. H. Nelson. 2003. Distributional and biological notes on Buprestidae (Coleoptera) in North and Central America and the West Indies, with validation of one species. The Coleopterists Bulletin 57(1):57–70.
Nelson, G. H., and T. C. MacRae. 1990. Additional notes on the biology and distribution of Buprestidae (Coleoptera) in North America, III. The Coleopterists Bulletin 44(3):349–354.
Copyright © Ted C. MacRae 2010